Sooner or later, many fluid power designers will face the problem of a hydraulic or pneumatic system that seems impossible to seal. Maybe the system’s temperature is extremely high. Or maybe its pressure is way above normal. Whatever the cause, these unusual environments have the potential to reduce the average familiar black and round seal to a small heap of extruded carbon molecules.
With today’s materials technology, sealing unusual high-performance applications doesn’t have to be a headache. Following a few simple guidelines can reduce your design time and your seal-failure rate — not to mention your blood pressure.
High differential pressure
Four basic questions must be answered before attempting to specify seals for high-pressure applications:
• What is the differential pressure across the seal?
• How large is the radial extrusion gap between the bore and the rod or piston?
• Is the seal application static or dynamic?
• What temperatures will the seal see?
Although system pressure of an application is easier to find, the differential pressure across the seal is what really matters for sealing. Many accumulator seals see very high pressure, but their differential pressure is very low because pressure is approximately equal on both sides. Differential pressure can be calculated simply by subtracting the pressure on one side of the seal from the pressure on the other side.
High differential pressure works to extrude a seal through any available gap. The material directly blocking the gap will be under the most stress. The key to sealing high pressures is to place a material with a modulus of elasticity higher than that of the seal directly over the extrusion gap.
High-modulus materials often are found in backup rings, but they also can be used as the seal material itself. Parker compounded Resilon ER (formerly known as 4304) with a high modulus comparable to that of traditional backup materials (such as Parker’s Polymyte and Fluoromyte), but Resilon ER also has the high abrasion resistance and resilience characteristics of PPDI polyurethane.
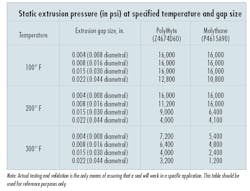
It is difficult to rate seal materials for a certain differential pressure, because that rating is affected by the extrusion gap, the motion, and the temperature. (In addition, seals in static applications usually will be able to contain two to three times as much pressure as the same seals under the same conditions when dynamic motion is introduced.) Higher temperatures lower the material’s modulus of elasticity and, consequently, its resistance to extrusion, Figure 1.
When calculating radial extrusion gaps, it’s tempting to simply divide the diametral clearance in half. But results obtained by this method may be misleading. Often, side loading will push the piston or rod over to one side and leave most of the clearance on the other. Wear rings can complicate this calculation even further due to non-concentricity and stack-up tolerances.
The results from static pressure testing on two of Parker’s proprietary seal materials are shown in the table. (Note that dynamic extrusion resistance will likely be much lower.)
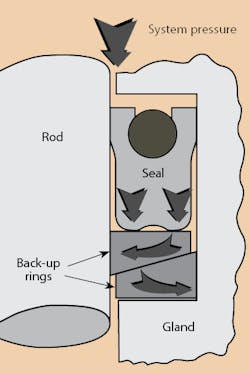
Installing backup rings made of poly-ether-ether-ketone (PEEK) or brass is one way to deal with extremely high pressures. To close the extrusion gap mechanically, use a positively actuated backup. These backups are placed in the gland in such a way that they are pushed closer to the sealing surface by the downward force exerted by pressure, Figure 2. The higher the pressure, the greater the force that holds them against the sealing surface and in front of the extrusion gap. Positively actuated brass backup devices have contained pressures exceeding 90,000 psi under static conditions. Their 0.025-in. radial clearance proved insignificant.
Because relatively few designers confront very high pressures regularly, the old adage, “If you feel like you are getting in over your head, you probably are,” certainly applies here. If you are unsure about a high-pressure situation, the best thing to do is to answer the four basic questions at the start of this section and then call the factory applications engineers for help.
Extreme temperatures
When choosing a seal for a high-temperature application, only two basic questions must be answered:
• What fluids will the seal contact?
• What will the contact temperature be?
It’s pretty safe to assume that some reaction will occur between any two dissimilar materials if their temperature gets hot enough. This assumption certainly holds true in the sealing industry. A question about a seal’s chemical compatibility cannot be answered without knowing the temperature involved, and a question about a seal’s temperature limit cannot be answered without knowing the fluid the seal will contact. Both factors must be considered in tandem.
This makes it difficult to assign a temperature limit to a seal material. At Parker Packing, we try to be practical about temperature limits of materials by assuming the seal is exposed to the fluid of its most common usage. In other words, polyurethane is temperature-rated for hydraulic oil, not water. Ethylene propylene is temperature-rated in water-glycol fluid, not petroleum-based hydraulic oil.
Some seal materials do not react with much of anything until the temperature gets very hot. Among these high temperature materials are polytetrafluoroethylene (PTFE), HiFluor, Parofluor, and PEEK. Although PTFE has little elasticity and must use mechanical spring energizers to facilitate its sealing capability, it is the most commonly used of these high-temperature materials. HiFluor and Parofluor, both from the same family of fluorinated elastomers, are expensive, but some applications call for their superior sealability and chemical resistance at high temperatures. PEEK normally is not used as an actual sealing element, but it makes an excellent anti-extrusion material due to its high modulus of elasticity.
At the opposite end of the temperature scale, many elastomers lose their elasticity at temperatures below –20° F. They become hard and brittle when the temperature approaches the material’s glass-transition temperature (the point at which the molecules rigidly attach themselves together). The elastomer usually is not damaged at these low temperatures, but it no longer has the resilience to push back against the surrounding surfaces to form a seal.
PTFE is also very good at cold temperatures. (In the extreme, Parker’s Polon 110 PTFE actually has been known to maintain flexibility down to –450° F.) For more common low-temperature applications, there is always ethylene propylene and polyurethane; both polymers will seal to –65° F.
Fluid considerations
Knowing exactly what is in an exotic fluid obviously is an important part of the process of selecting seal material for it. Material Safety Data Sheets (MSDSs), usually available from the fluid supplier, are very helpful when trying to identify the more hazardous components of a fluid. If you know the relative concentrations of each fluid component, you’ll often be able to spot compatibility problems up front. However, while certain acids may be listed as incompatible with certain seal materials, there may be no noticeable reaction if the concentration of that acid is low.
Note that the purpose of an MSDS is to focus on what is potentially dangerous to humans, not to seal materials. Water will never be listed among the hazardous components on an MSDS, but water certainly will cause polyurethane to degrade — molecule by molecule. Even though fluid components that are harmless to humans will not appear on the MSDS, their effect on seal materials still must be considered.
Epilogue
New equipment designs and processes constantly challenge the craftsmanship of seal-material chemists. The market calls for higher pressure capabilities to allow for more compact components. Elevated-temperature processes call for elastomeric seals to perform in situations once thought impossible. The future surely holds new advances in chemistry for tomorrow’s extreme applications. And it will be the design engineer’s challenge to select the right seal material and configuration for those applications.
Mike Thomas is application engineer, Parker Packing Div. (now the Engineered Polymer Systems Div.), Salt Lake City. For more information, call (801) 972-3000 or visit www.parker.com/eps.